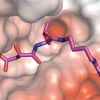
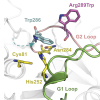
Structural biology is used to determine the shape of molecules at atomic resolution. This allows us to understand exactly how disease mutations affect proteins and how we can correct them. An important goal in the lab is to understand the structure of Calpain-5 (CAPN5), a calcium-activated regulatory cysteine protease that is expressed throughout the central nervous system and retina. Mutations in the catalytic core of CAPN5 make it hyperactive and cause Autosomal Dominant Neovascular Inflammatory Vitreoretinopathy (ADNIV; OMIM#193235), a severe, blinding eye disease. Structural studies of CAPN5 ADNIV disease mutants will inform how these mutations alter CAPN5 structure and function. Our laboratory is using x-ray crystallography and other biophysical techniques (small angle x-ray scattering, dynamic light scattering, cryo-EM, and fluorescence assays) to study the structural mechanisms of CAPN5 activation.
The lab also uses molecular modeling to study disease-causing mutations. With the rapidly-reducing costs of whole genome and exome sequencing, clinical genomic testing is being increasingly applied in the identification of pathogenic disease variants in patients. New sequence variants are often identified, but many are classified as variants of uncertain significance (VUS) due to the lack of functional assays to study their effects. Molecular modeling of protein structures can provide information at the atomic level and help to predict the pathogenicity of novel mutations by placing variants of uncertain significance in the context of the patient’s disease, pathophysiology, and protein function. Our team collaborates with clinicians and researchers around the world to model novel disease variants and better understand their patients’ disease. Please contact us for a consultation or collaboration.